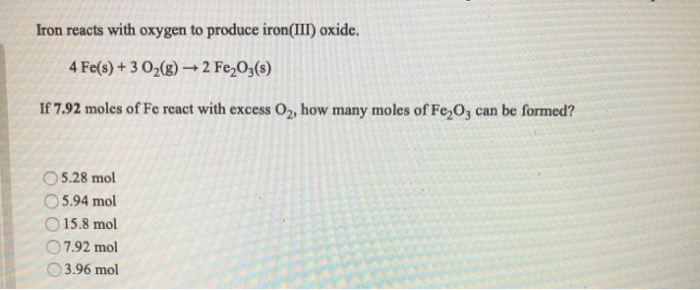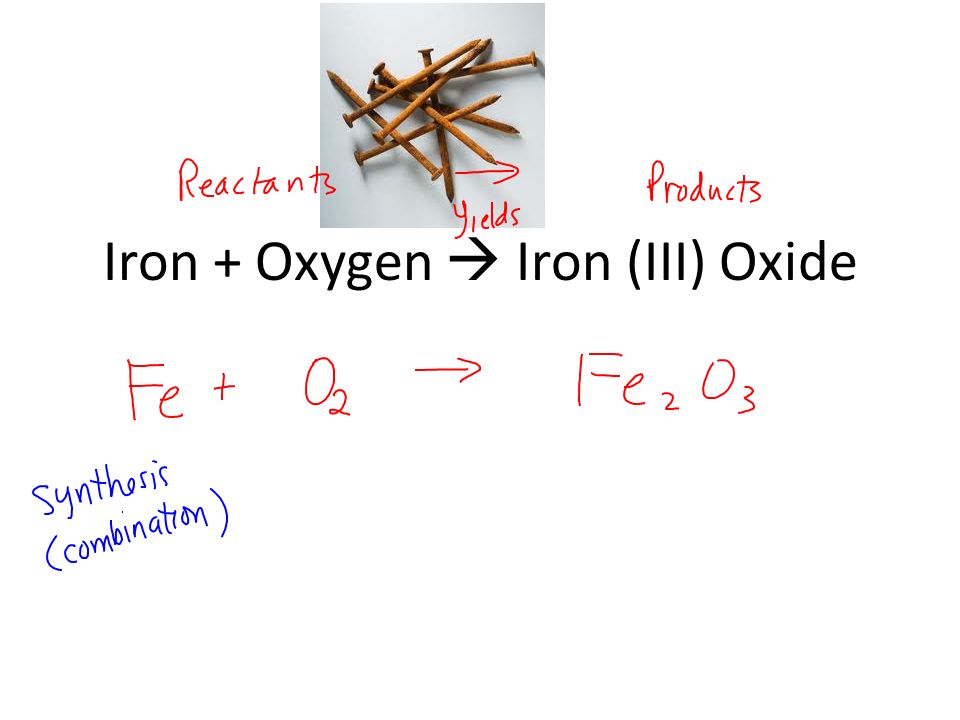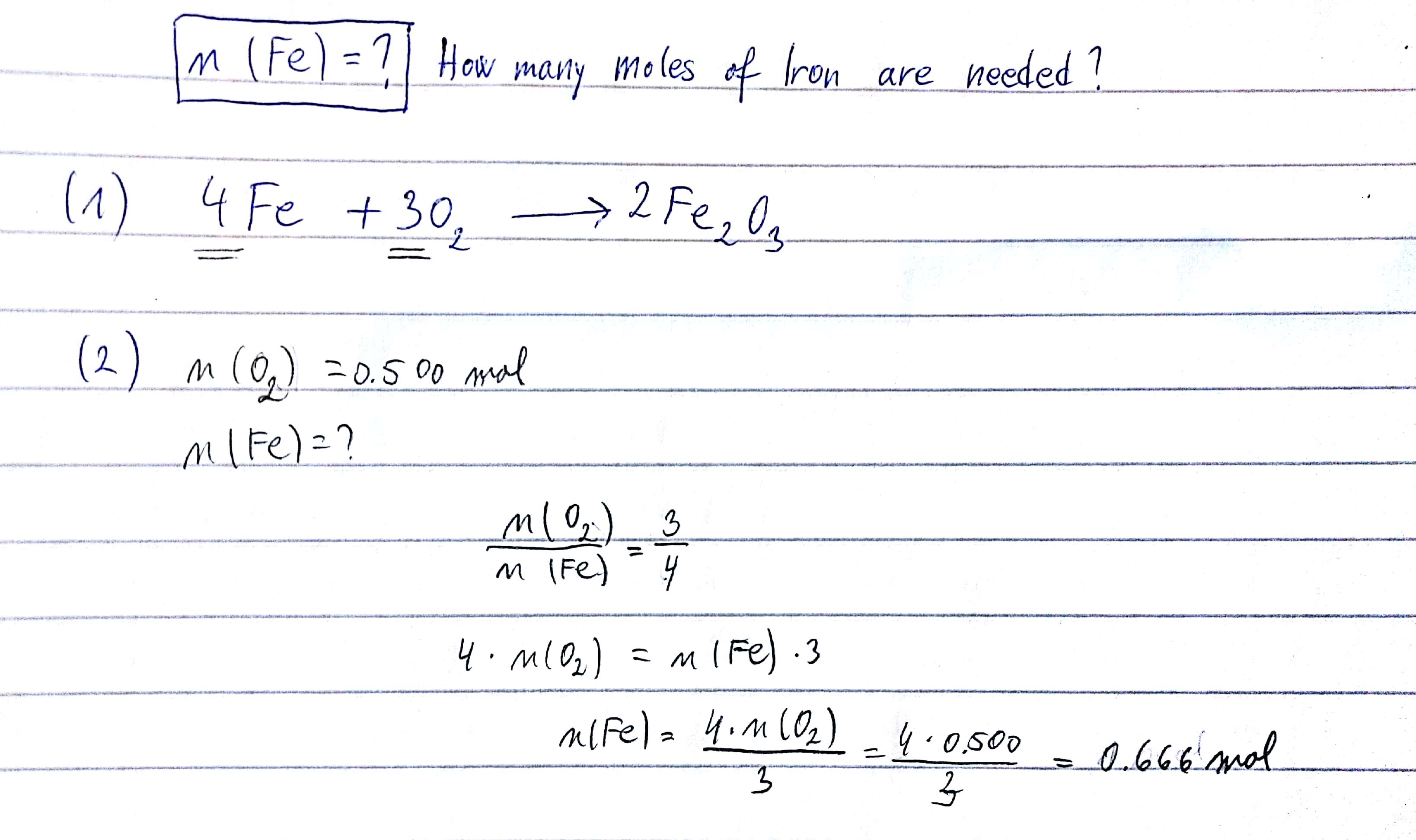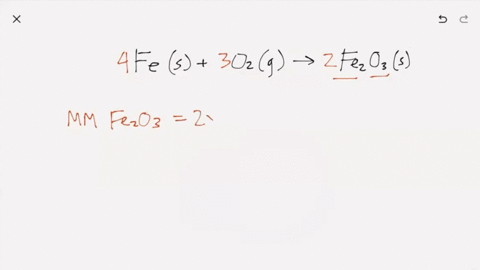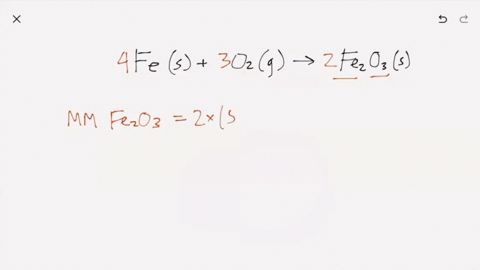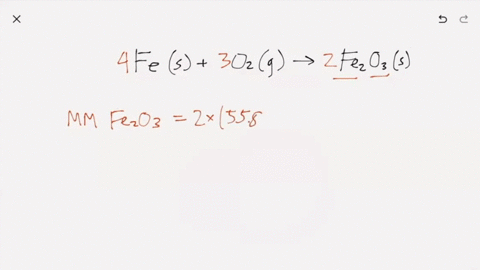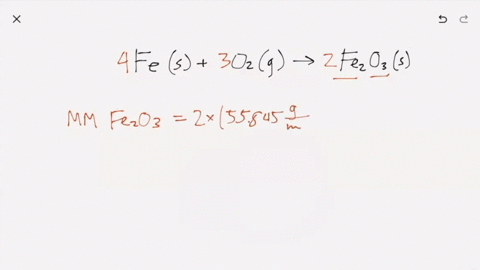
SOLVED:Iron metal reacts with oxygen to give iron(III) oxide, Fe2 O3 (a) Write a balanced equation for the reaction. (b) If an ordinary iron nail (assumed to be pure iron) has a

⚗️Iron (Fe) reacts with oxygen gas (O2) to form rust (Fe2O3). Balance the equation below by writing - Brainly.com

Vinegar And Clr Are My New Best Friends - Iron Rusting Chemical Equation PNG Image | Transparent PNG Free Download on SeekPNG
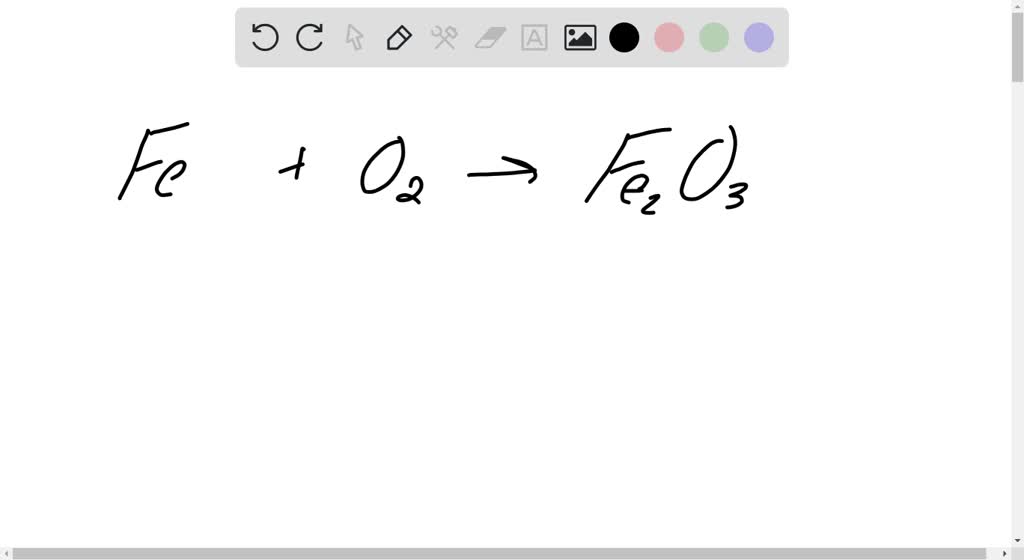
SOLVED: Write a balanced chemical equation based on the following description: iron metal reacts with oxygen gas to produce solid iron(IlI) oxide
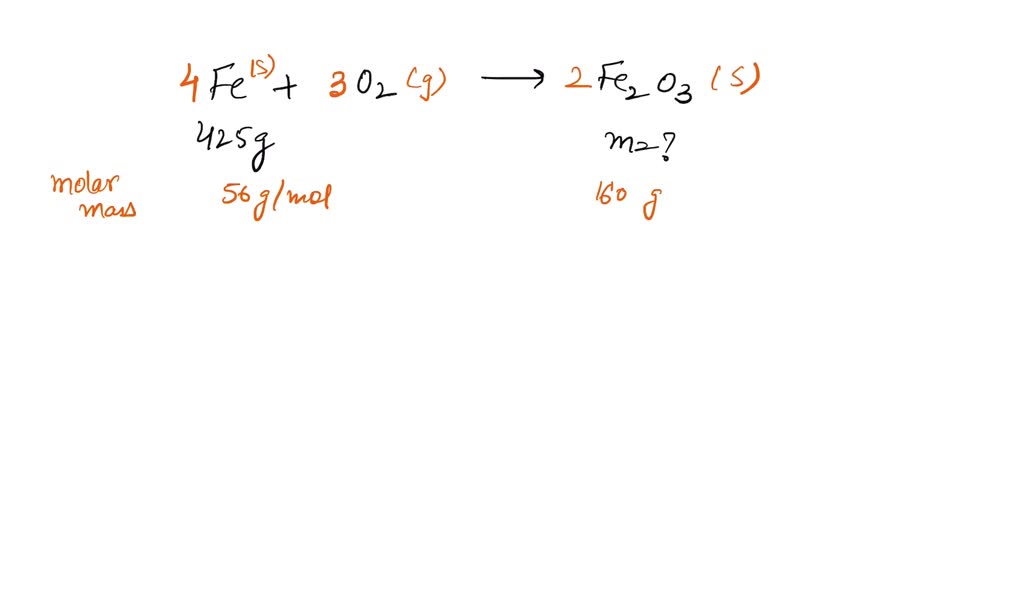
SOLVED: The formation of rust which is iron (III) oxide on the surface of iron metal is an oxidation-reduction reaction between iron metal and oxygen gas. balanced equation : Fe (s) +